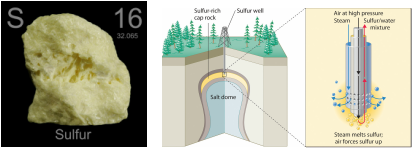
To create sulphuric acid, you will primarily need sulfur. Once you have located a source of sulfur, you should pump hot water into the ground, which causes the sulfur to melt. Once this is done, the molten sulfur is forced up out of the ground to cool. The next step is for it to be sent to a manufacturer. Once there, it can be created in two ways. One is through the lead chamber process and the through the contact process.

The contact process:
The contact process is quite similar to the lead chamber process. The first step is to heat the sulfur into oxygen, which forms sulfur dioxide. The next step is to heat the sulfur dioxide into oxygen, to form sulfur trioxide. The final step is to dissolve the sulfur trioxide into concentrated sulphuric acid, which forms oleum, a substance that is safe to transport and store. However, when more sulphuric acid is need, just add water to the oleum.
The contact process is quite similar to the lead chamber process. The first step is to heat the sulfur into oxygen, which forms sulfur dioxide. The next step is to heat the sulfur dioxide into oxygen, to form sulfur trioxide. The final step is to dissolve the sulfur trioxide into concentrated sulphuric acid, which forms oleum, a substance that is safe to transport and store. However, when more sulphuric acid is need, just add water to the oleum.
The lead chamber process:
This method is done in large lead-lined containers. The first step is to heat up the sulfur, which creates sulfur dioxide. The next step is to convert sulfur dioxide to sulfur trioxide. The last step is to add water to sulfur trioxide, which creates sulfuric acid.
This method is done in large lead-lined containers. The first step is to heat up the sulfur, which creates sulfur dioxide. The next step is to convert sulfur dioxide to sulfur trioxide. The last step is to add water to sulfur trioxide, which creates sulfuric acid.
